Section 7.4 – Method Verification
Introduction
A new analyzer (Diasorin Liaison) was aqcuired by the Virology Department at GSH.
In summary: since the Diasorin Liaison has the capability to do various chemistry analyses too, we were able to “piggy back” on the analyzer’s acquisition and install / setup various chemistry analytes too.
We have decided to introduce:
- Aldosterone
- Renin
- IGF-1
- Growth Hormone
- C-peptide
I was given the task to specifically concentrate on aldosterone as method verification.
Points which will be discussed:
- Rationale for implementation of Aldosterone and Renin
- Medical decision limits
- EP15-A3: 5×5 method for precision (and accuracy) determination
- Method comparison study
Rationale for implementation of Aldosterone and Renin
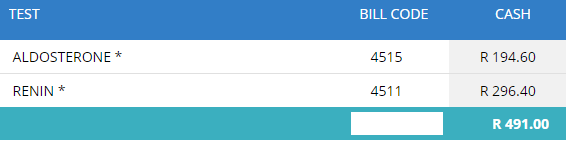
Our previous practice was to send aldosterone and renin results to a private laboratory (PathCare).
Virology had planned to budget for a new analyser to run a few of their Virology serology assays. Their test volumes, for the assays on their analyser hasn’t been high enough to adequately motivate for this new analyser.
The auto-analyser which we use (Roche COBAS 6000), doesn’t have a repertoire of Aldosterone and Renin.
Since we had already sent roughly 30 samples for both aldosterone and renin to Pathcare per month, this was a great opportunity to partner with the Virology department in a mutual tender for an instrument which could run their serology assays and our Aldosterone and Renins. Additionally we added some other tests to the repertoire for tender of tests which we were also sending to Pathcare: IGF-1 and hGH.
Together with the anticipated virology serology tests, the total test volume was such that we could motivate to the business manager at GSH for a “break-even” scenario with regards to Cost vs. Income.
The tender was then awarded to the company supplying the Diasorin Liason analyser (as a placement option for 5 years), upon which I was given the task of doing the method verification for aldosterone.
The prior CLSI EP15 A2 document described the following:
Clinical and Laboratory Standards Institute (CLSI) document EP05-A2 describes the protocols for determining the precision of a method. The precision of a method should be tested at at-least two levels; each run in duplicate, with two runs per day over 20 days. It also describes the protocols that should be undertaken by the user to verify precision claims by a manufacturer. Precision claims by a manufacturer should be tested at at-least two levels, by running three replicates over five days.
Excerpt – CLSI EP15 A2 2
Medical decision limits
The medical decision limits chosen were:
EP15-A3: 5×5 method for precision (and accuracy) determination
In the NHLS, a method verification currently happens according to guidelines as per CLSI EP15-A3 document: “User Verification of Precision and Estimation of Bias” provides a simple experimental approach to estimate a method’s imprecision and bias. 1
The objective is to determine if the laboratory precision performance of repeatability and within-laboratory imprecision are in accordance to the manufacturer specification claims.
Aldosterone was performed on the Diasorin Liason. One QC lot was obtained for QC of the assay after maintenance and another QC lot (with a different assigned value) was used as the testing material. Quality control (QC) levels (L1: 5.25; L2: 21.10 ng/dL) (Bio-Rad Laboratories, Irvine, CA) were used. Each QC level was measured before and after instrument. Five replicates of the testing material was done per run, one run per day, and during 5 days. Imprecision estimates were calculated by one-way analysis of variance using Microsoft Excel on a template sheet developed previously by Dr Fierdoz Omar. Estimated imprecision was compared to the manufacturer’s stability claims and desirable imprecision specifications based on biological variation.
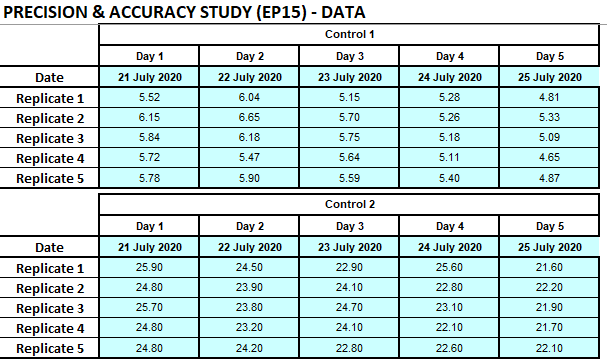
Result
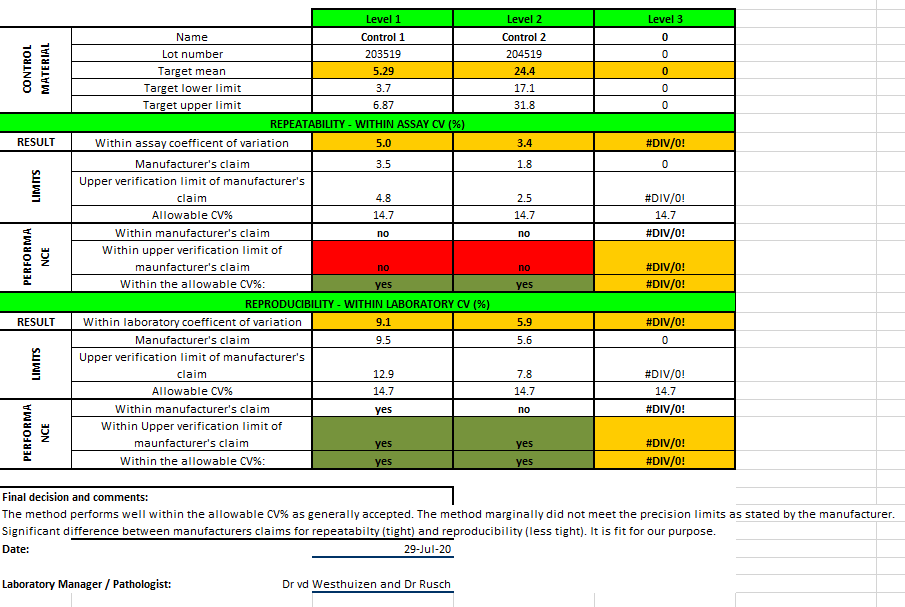
See below the attached verification report:
The full report can be downloaded here:
Conclusion
The aldosterone assay was found “fit for purpose” although not meeting all the manufacturer’s claims.
The literal description of “fit for purpose”: (of an institution, facility, etc.) well equipped or well suited for its designated role or purpose.
This assay, even though not meeting the manufacturer’s claims, performs well within the published “Desirable Analytical Quality Specifications for Imprecision, Bias and Total Error Upon Biological Variation”.
In the process of this project and every day IQC, I have also developed an interactive portal with the desirable analytical quality specifications as per Ricos et al.: TEa Dashboard
References
- Jose Jara Aguirre, MD, Karl Ness, MLS, Alicia Algeciras-Schimnich, Application of the CLSI EP15-A3 Guideline as an Alternative Troubleshooting Tool for Verification of Assay Precision, American Journal of Clinical Pathology, Volume 152, Issue Supplement_1, October 2019, Page S88, https://doi.org/10.1093/ajcp/aqz117.007
- Chesher D. Evaluating assay precision. Clin Biochem Rev. 2008 Aug;29 Suppl 1(Suppl 1):S23-6. PMID: 18852851; PMCID: PMC2556577, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2556577/
- Desirable Analytical Quality Specifications for Imprecision, Bias and Total Error Upon Biological VariationDerived from Ricos C, Alvarez V, Cava F, Garcia-Lario JV, Hernandez A, Jimenez CV, Mininchela J, Perich C, Simon M. “Current databases on biologic variation: pros, cons and progress” Scand J Clin Lab Invest 1999;59:491-500. Updated from data made available in 2014.Source: https://www.qcnet.com/Portals/0/PDFs/BVValues1Final.pdf