The Vitamin D conundrum
| HOSP # | WARD | F22 Orthopaedics Ward | |
| CONSULTANT | Dr. Jody Rusch | DOB/AGE | 42 Y Female |
Abnormal Result
Total Vitamin D of 27.1 nmol/L on 18 March 2020.
Total Vitamin D of 65.4 nmol/L on 01 April 2020.
Presenting Complaint
Patient had a low impact femur fracture on 18 March 2020 :
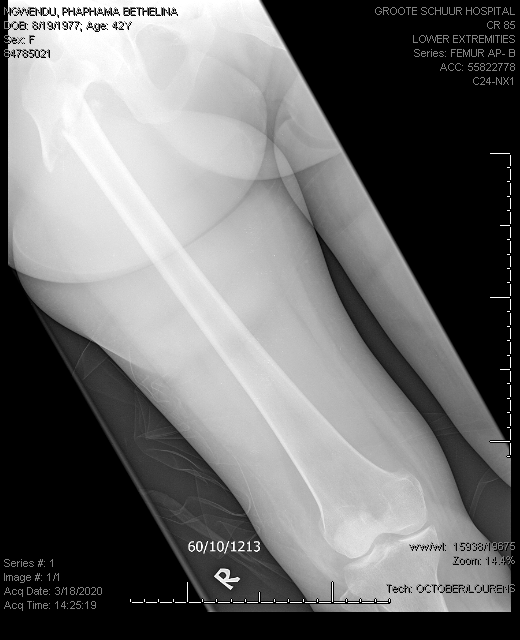

History
Patient is known with:
- previous deep venous thrombosis in 2018, on Warfarin therapy
- ?Epilepsy, patient is on carbamazepine, for which the Endocrinology specialists were of opinion that it may have been the cause of the low Vitamin D level.
Examination
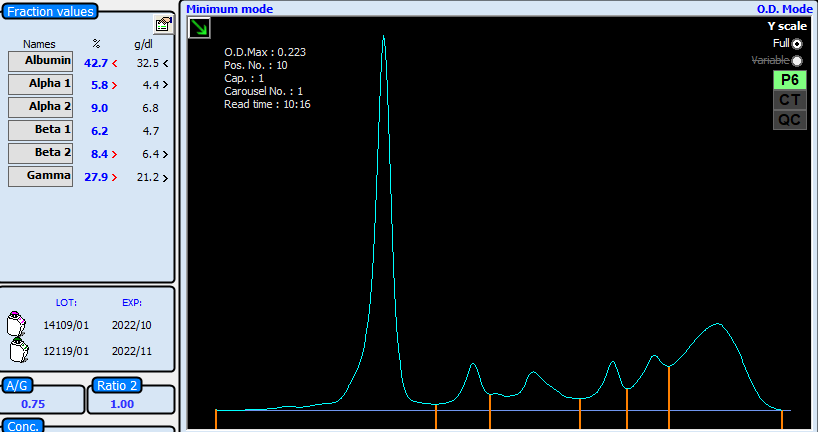
Laboratory Investigations
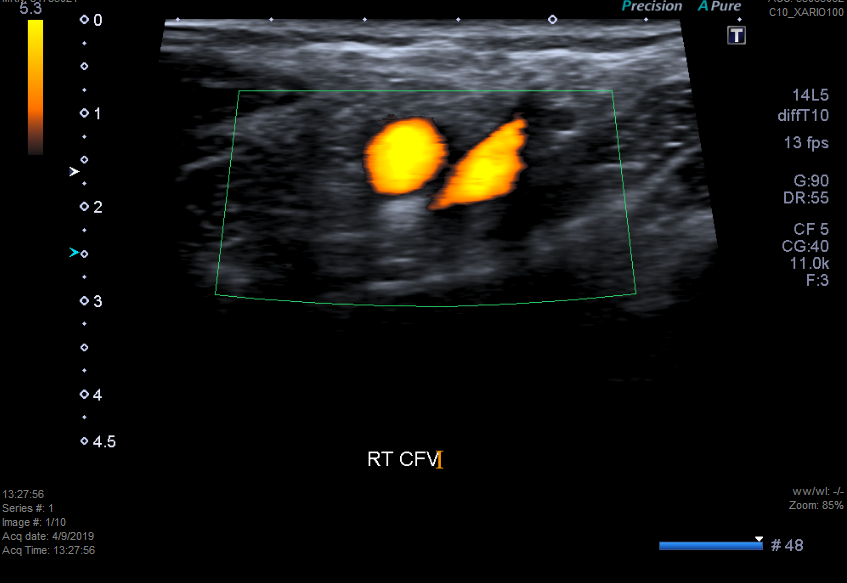
Other Investigations
Final Diagnosis
Vitamin D deficiency likely due to carbamazepine therapy.
Take Home Message
I was not aware that patients on carbamazepine (or other enzyme inducing agents) have lower Vitamin D levels, and it became evident after a quick literature search that it was in fact the case, see the abstract of the article below, also see another article written by a colleague of mine, Jusine Cole, on the Vitamin D controversies.
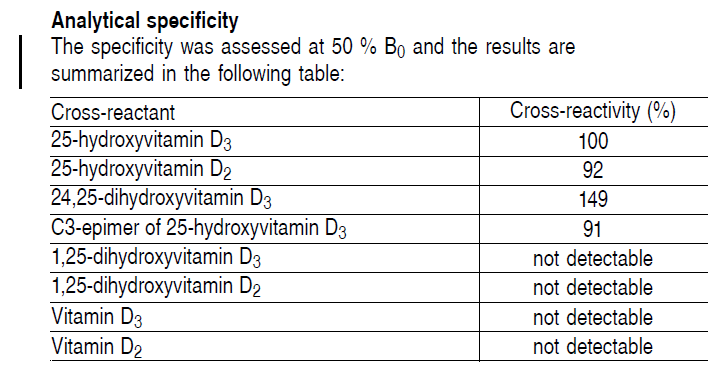
Also, I have learned that although “total Vitamin D”, as the assay is named in our immunoassay package insert and on TrakCare LIS, has to do with the total portion with regards to protein binding (to Vitamin D binding protein) and not so much to the fact that calcitriol and calcidiol is measured.
It is however evident that, since the cross-reaction in the immuno-assay is quite pronounced with the various forms of Vitamin D, that total indeed, might be an accurate description. In reality, the assay is however called the Total 25-hydroxy Vitamin D.
Vitamin D levels and bone turnover in epilepsy patients taking carbamazepine or oxcarbazepine.
Abstract
PURPOSE:
Evidence suggests that enzyme-inducing antiepileptic drugs (AEDs) may decrease serum 25-hydroxyvitamin D (25-OHD) levels and increase bone turnover. We sought to determine whether these are affected by treatment with carbamazepine (CBZ) or oxcarbazepine (OXC).
METHODS:
We measured serum levels of 25-OHD, parathyroid hormone (PTH), osteocalcin (OCLN), bone alkaline phosphatase (BAP), and urinary N-telopeptides of type I collagen cross-links (NTX) in normal controls (n=24) and in epilepsy patients taking CBZ (n=21) or OXC (n=24) in monotherapy. CBZ patients were subsequently switched overnight to OXC monotherapy, and after 6 weeks, the tests were repeated.
RESULTS:
25-OHD levels were lower in each drug-treated group (OXC, 19.4+/-2.3 pg/ml; CBZ, 20.4+/-2.4) than in the controls (27.5+/-2.8) (ANOVA, p=0.052). This difference was significant for the OXC group (p<0.05). PTH, BAP, and NTX did not differ significantly among groups. OCLN levels were somewhat elevated in the OXC group (2.79+/-0.47 ng/ml) and more clearly and significantly elevated in the CBZ group (3.63+/-0.36) compared with controls (2.38+/- 0.41) (p=0.053). Because the data were very similar between OXC and CBZ groups, they were combined to increase statistical power. The combined drug-treatment group had significantly higher BAP (p=0.02) and lower 25-OHD (p=0.015) than did controls. The latter remained significant even after accounting for the confounding effects of age on 25-OHD levels (p<0.05). No significant differences were found after CBZ patients were switched to OXC.
CONCLUSIONS:
Epilepsy patients taking OXC or CBZ have significantly lower 25-OHD than do normal controls, with a pattern of changes in other bone biomarkers suggestive of secondary hyperparathyroidism. It may be prudent for patients taking CBZ or OXC to be prescribed 25-OHD replacement.

