A possible case of growth hormone deficiency
| HOSP # | WARD | G4 Adolescent Ward | |
| CONSULTANT | Ariane Spitaels / Heleen Vreede | DOB/AGE | 12 y Male |
Abnormal Result
Low growth hormone with an abnormal clonidine stimulation test.
Presenting Complaint
At 7 years of age, patient presented with chronic headaches.
History
CT-brain revealed acute hydrocephalus. Endoscopic 3rd ventriculostomy was done. MRI revealed a lesion posterior to mid-brain and pons as well as a hyper-intensity on T1 with contrast. A glioma was queried as the cause.
Patient recently was identified to have decreased growth velocity (4cm / year) in the preceding 6 months and was also admitted for a clonidine stimulation test. Patient was primed for 2 days.
Examination
N/A
Laboratory Investigations
Histology
GROSS DESCRIPTION: Specimen labelled Sub-occipital tumour consists of 5 fragments of tissue measuring 4-5mm each.
HISTOLOGY: Sections of the brain biopsies show sheets of bipolar neoplastic cells with piloid cytoplasmic processes. Scattered pleomorphic tumour cells, microcystic areas and areas resembling oligodendrocytes are present. Rosenthal fibres and eosinophilic granular bodies are not a prominent feature. There is no vascular proliferation or tumour necrosis.
Comment: See frozen section below.
PARAFFIN SECTIONS: Sections of the brain tumour show piloid areas (cells with hair-like processes, gemistocytic cells and focal Rosenthal fibres. Mild nuclear atypia is noted. There are no mitotic figures, vascular proliferation or areas of necrosis. Scattered entrapped neurons are noted.
IMMUNNOHISTOCHEMISTRY: Neurofilament: Highlight background neural processes
GFAP: Diffusely positive p53: Weak staining in less than 10% of the tumour cells, interpreted as negative.
Ki67 proliferation index: < 4%
CONCLUSION: Brain, biopsy: Juvenile pilocytic astrocytoma.
Other Investigations
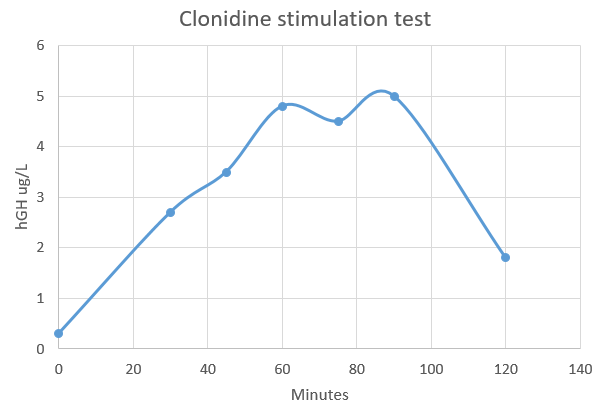
Clonidine stimulation testing
| Time after Clonidine injection (minutes) | Result |
| 0 | 0.3 |
| 30 | 2.7 |
| 45 | 3.5 |
| 60 | 4.8 |
| 75 | 4.5 |
| 90 | 5 |
| 120 | 1.8 |
Final Diagnosis
Since the hGH concentration did not rise to above 10 ug/L, the diagnosis is likely that of growth hormone deficiency, or hyposomatotropism.
Take Home Message
Provocative Testing for Growth Hormone deficiency (GHD)
The following provocative (dynamic) tests are available for hGH deficiency:
- Insulin tolerance test
- Clonidine stimulation test
- Levodopa-propranolol HCl test
- Arginine HCl test
- Glucagon test
Provocative GH testing is criticized for several reasons, including the following:
- None of the tests reproduces the physiologic secretory pattern of GH because they involve the use of pharmacologic stimuli to indirectly assess physiologic GH production.
- Individual clinicians assign what are essentially arbitrary definitions for subnormal responses (ie. cutoffs for peak serum GH values)
- The reproducibility of provocative tests and GH RIAs is limited. Many pediatric endocrinologists apply other clinical criteria (eg, growth velocity Z score below -2) and do not perform provocative GH tests to diagnose GHD.
Despite limitations, provocative GH tests remain helpful ways to measure GH reserve. Pediatric endocrinologists use physiologic stimuli (eg, strenuous exercise, fasting, deep sleep) and pharmacologic stimuli (eg, clonidine, levodopa-propranolol, glucagon, arginine, insulin) to provoke GH secretion. The tests must be performed after overnight fasting, firstly ensuring that the children are euthytoid.
To improve diagnostic sensitivity and specificity, at least 2 provocative tests are performed. Immediately before and during the earliest phases of puberty, GH production is often indistinguishable in unaffected children and in children with GHD. Serum GH concentrations typically rise during puberty. Many investigators suggest that children approaching puberty should be given gonadal steroids to prime the growth hormone-releasing hormone (GHRH)-GH axis before testing.
Most clinicians use a peak serum GH concentration of more than 10 ng/mL (30 IU) to exclude GHD in children. Specific provocative tests are described below.
Insulin tolerance test
Insulin-induced hypoglycemia is the most potent stimulus for GH secretion and the most dangerous tool for provocative GH testing in patients who may have GH deficiency. Insulin tolerance testing takes advantage of the hormonal counterregulatory response to hypoglycemia. In patients without GHD, plasma concentrations of glucagon, epinephrine, norepinephrine, cortisol, corticotropin, and GH are elevated in response to acute hypoglycemia.
To perform the test, patients fast for 8 hours. Then, lispro insulin 0.1 U/kg of body weight is administered rapidly as an intravenous bolus. Serial blood samples are subsequently obtained to measure GH, cortisol, and glucose concentrations at 0, 15, 30, 60, 75, 90, and 120 minutes. With each sample, the blood glucose level is simultaneously determined by using a bedside glucometer to document an appropriate reduction and to ensure safety. Performance of the test is considered adequate when the blood glucose level decreases below 50% of its baseline value.
Adverse effects expected during the procedure include symptoms secondary to hypoglycemia, such as lethargy, shaking, confusion, headache, abdominal pain, nausea, vomiting, syncope, and seizure activity. The test must be performed under the watchful eye of the physician who can begin prompt resuscitation with glucose and/or glucagon as soon as the diagnostic samples have been obtained. To date, the insulin tolerance test is the only provocative test associated with fatalities; therefore, personnel must be trained and conduct the test judiciously.
Clonidine stimulation test
Clonidine acts centrally to stimulate alpha-adrenergic receptors, which are involved in regulating GH release. Serum GH levels are obtained at baseline and at 60 minutes and 90 minutes after the oral administration of clonidine 0.1 mg/kg. Clonidine may induce hypotension during the test. Therefore, warn parents that they may experience lethargy and/or depression for 24 hours after clonidine is administered.
Levodopa-propranolol HCl test
Levodopa is a dopamine receptor agonist. Dopamine is involved in the stimulation of GH secretion. In the converse, beta-adrenergic control negatively regulates GH release.
Propranolol is a beta-blocker used to hinder inhibitory input affecting GH release, while levodopa simultaneously stimulates GH release by means of the dopaminergic pathway. Propranolol 0.75-1 mg/kg is orally administered before levodopa. The dosage of levodopa for levodopa-propranolol HCl testing varies with weight, so that children weighing less than 15 kg receive 125 mg, children weighing 10-30 kg receive 250 mg, and children weighing more than 30 kg receive 500 mg.
Blood samples for GH testing are drawn at 0, 60, and 90 minutes after the administration of levodopa. Adverse effects include nausea and, in rare cases, emesis. In addition, the patient’s heart rate may decrease because of the use of propranolol. Closely monitor his or her vital signs, and ensure that appropriate resuscitative measures are available.
Arginine HCl test
Arginine appears to exert a direct depolarizing action on somatropic neurons, increasing GH secretion. After an overnight fast, patients are given 10% arginine HCl in 0.9% NaCl 0.5 g/kg (not to exceed 30 g) as a constant intravenous infusion over 30 minutes. Blood samples for GH testing are obtained at 0, 15, 30, 45, and 60 minutes after the infusion of arginine is begun. Arginine has historically been used as a primer before insulin is administered during insulin tolerance testing.
Glucagon test
Glucagon increases peripheral glucose concentrations by means of glycogenolysis and gluconeogenesis. Because glucagon is rapidly metabolized, an abrupt reduction in serum glucose concentration ensues and triggers the release of counterregulatory hormones.
After fasting overnight, patients receive an intramuscular injection of glucagon 0.03 mg/kg (not to exceed 1 mg). Some clinicians advocate the concomitant use of propranolol to inhibit the catecholaminergic response to hypoglycemia. Serum GH concentrations are determined at 0, 30, 60, 90, 120, 150, and 180 minutes after glucagon administration. Nausea and, occasionally, emesis may occur.
Growth hormone reference intervals provided should be seen as a guideline only. Specifically, reference intervals or random growth hormone levels are not useful for diagnosing growth hormone deficiency.
For this, stimulation tests are required. A recent paper established clinical evidence-based cut-off limits for
diagnosing growth hormone deficiency in children and adolescents following growth hormone stimulation tests, by various commercially available immunoassays, compared to isotope dilution mass spectrometry (ID-MS).
Reference: Clinical evidence-based cutoff limits for GH stimulation tests in children with a backup of results with reference to mass spectrometry, Wagner et al, European Journal of Endocrinology (2014)171, 389-397TrakCare Autocomment
| Assay | Cut-off limit |
| Immulite 2000 (Siemens) | 7.77 ug/L |
| AutoDELFIA (Perkin-Elmer) | 7.44 ug/L |
| iSYS (Immunodiagnostic System) | 7.09 ug/L |
| Liaison (DiaSorin) | 6.25 ug/L <– Current assay |
| DXI (Beckman-Coulter) | 5.15 ug/L |