Anti thyroid hormone resistance
| HOSP # | WARD | Obstetrics | |
| CONSULTANT | Dr. Khalid / Dr. Jody Rusch | DOB/AGE | 27y female |
Abnormal Result
TSH<0.01 IU/L, Free-T4 53.1 pmol/L Free-T3: 13.8 pmol/L
Presenting Complaint
G2 P1 GA 30 wks, presented with a large thyroid gland.
Was admitted with uncontrolled hyperthyroidism and congestive cardiac failure.
ECHO: RHD, severe MR, severe TR, EF = 56%
Foetal U/S: normal singleton pregnancy matching with gestational age & no obvious congenital malformations
History
Patient was diagnosed with hyperthyroidism in 2019
Received interrupted courses of NMZ (20 to 30 mg)
Now she is on regular 30 mg NMZ since April 2020, which increased to 40 mg and then to 60 mg in March 2021
Examination
GCS 15/ 15
BP 126/ 63 Pulse 99
Enlarged thyroid mostly Rt lobe with bruit, no eye signs, no enlarged LNs
Had gallop, pan systolic murmur, basal creps and lower leg edema
Laboratory Investigations
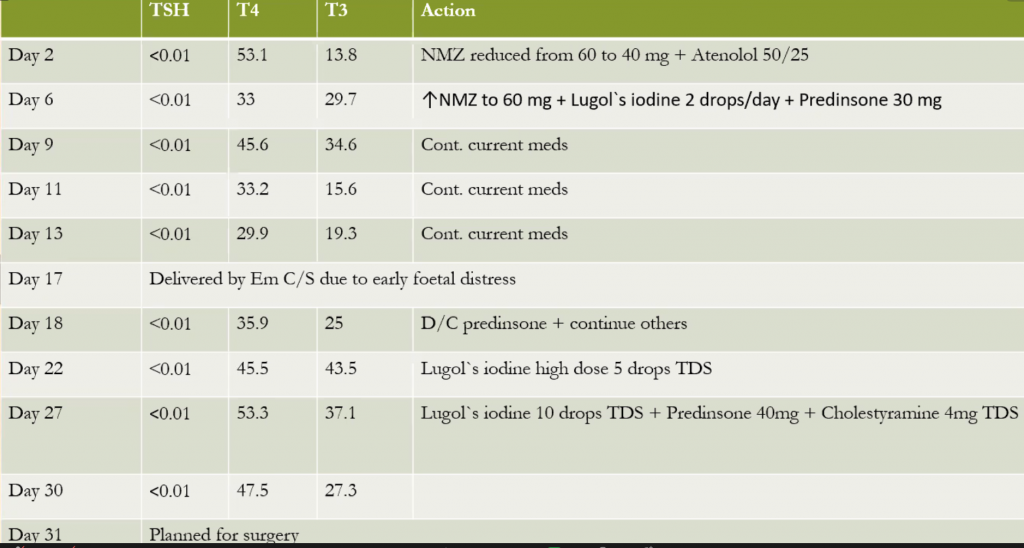
Other Investigations
Chest X-ray and CT-chest revealed –> No PE, tracheal narrowing.
U/S thyroid -> A large isoechoic nodule, occupying the whole RT lobe, TIRAD 3.
FNAB RT nodule -> Consistent with cyst, Bethesda 1.
Final Diagnosis
Resistance to antithyroid drugs
Take Home Message
ATDS Resistance, Pregnancy & Lactation
- Antithyroid drugs (ATDS) have been used in clinical practice for more than half a century
- ATDs side effects occur in 3%—5% of patients, the majority of which are allergic reactions such as skin rash, whereas the severe side effects of agranulocytosis (0.15%) and liver failure (<0.1%) are rare.
- Birth defects occurs in 2%—4% of children who have been exposed to MMI in early pregnancy, especially during gestational weeks 6-10.
- MMI, PTU, & CM, all cross the placenta, and can modulates fetal thyroid function. Importantly, all ATDs tend to be more potent in the fetus than in the mother. Thus, when the mother is made euthyroid, the fetus is often over treated.
- Therefore, the aim of treatment is to use the smallest possible dose to maintain maternal TT4/ FT 4 values at, or just above the pregnancy—specific ULM.
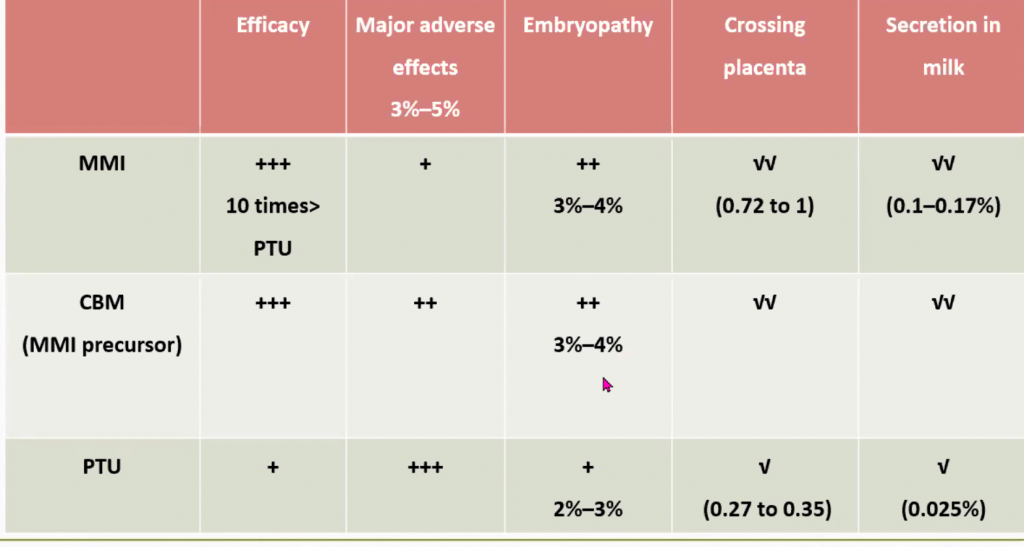
Effects of ATDS on pregnancy:
- Drug or class effect
- Daily or cumulative dose
- Time and duration of exposure
- Maternal thyroid function
Timing and type of treatment
A subset of the observational studies performed included a group of children considered as exposed to both MMI and PTU in earlv pregnancy. Half of the studies reported that such ‘double exposure’ was associated with a higher risk of birth defects as compared to non-exposed.
A more detailed evaluation of double exposed cases has indicated that the duration of MMI exposure in early pregnancy is critical. This finding may favor a shift in therapy prior to pregnancy also given that the critical window of exposure is from pregnancy week 6 to 10.
Maternal Thyroid Function
A small subset of observational studies on the use of ATDs and birth defects
provided data on maternal thyroid function in pregnancy.
– However, in one of the cohorts, maternal overt hyperthyroidism was a risk factor for birth defects in the child (adjusted HR: 1.91).
-In the Japanese study (2012), it was also reported that the frequency of birth defects was higher in hypothyroid women compared to euthyroid women.
-In another report from Japan sowed a rate of fetal malformations of:
- 0% (0 of 126) in treated euthyroid women
- 1.7% (2 of 117) in treated hyperthyroid women
- 6% (3 of 50) in untreated hyperthyroid women
Anti-thyroid Drug Resistance
Sporadic cases of resistant thyrotoxicosis are reported.
Etiologies of resistant thyrotoxicosis in literature include type I amiodarone induced thyrotoxicosis (AIT) and Graves disease.
Refractory cases have mostly shown resistance to high dose thionamides and beta-blockers; rarely resistance to iodine has also been reported.
Possible mechanisms responsible for resistance to ATDs include:
- Noncompliance is the most likely culprit
- Drug malabsorption
- Rapid drug metabolism
- Antidrug antibodies
- Impaired intrathyroidal drug accumulation or action
- Predominant elevation of T3 rather than T4 levels
Workup of Patients with Resistant Thyrotoxicosis.
- Evaluation of patient compliance.
- Urinary iodine excretion to exclude iodine contamination
- hilalabsorption of the drug: careful history taking and physical examination.
- Measurement of drug levels and plotting in a normal concentration-time curve
- Measurement of anti-drug antibodies (Recovery test I incubation with Protein-G)
- Intrathyroidal ATDs concentrations (high pressure liquid Chromatography).
- Resistance to drugs can be tested by performing a perchlorate discharge test
- Thyroid peroxidase activity.
Perchlorate discharge test
Perchlorate inhibits NIS function (sodium iodine symporter) eliminating the iodine gradient which is required for maintaining the iodine in the gland. This will results in a partial discharge of radiolabelled iodide from the thyroid indicating an impaired organification.
- A test dose of radioiodine administered first, and then 2 hrs later Potassium perchlorate or
thiocyanate is administered. - Perchlorate or thiocyanate is a competitive inhibitor of iodine transport into thyrocyte.
- In normal individuals in whom the organification and coupling remain intact only <10% of radioiodine is discharged (leaked), when iodine transport is inhibited.
- In individuals with defective organification and coupling, 40-90% of radioiodine is discharged (leaked), when iodine transport is inhibited.
Treatment of resistant thyrotoxicosis
Resistant thyrotoxicosis is managed by definitive treatment options: surgery or radioactive iodine ablation after euthyroidism is achieved by ATDs to minimize perioperative adverse events.
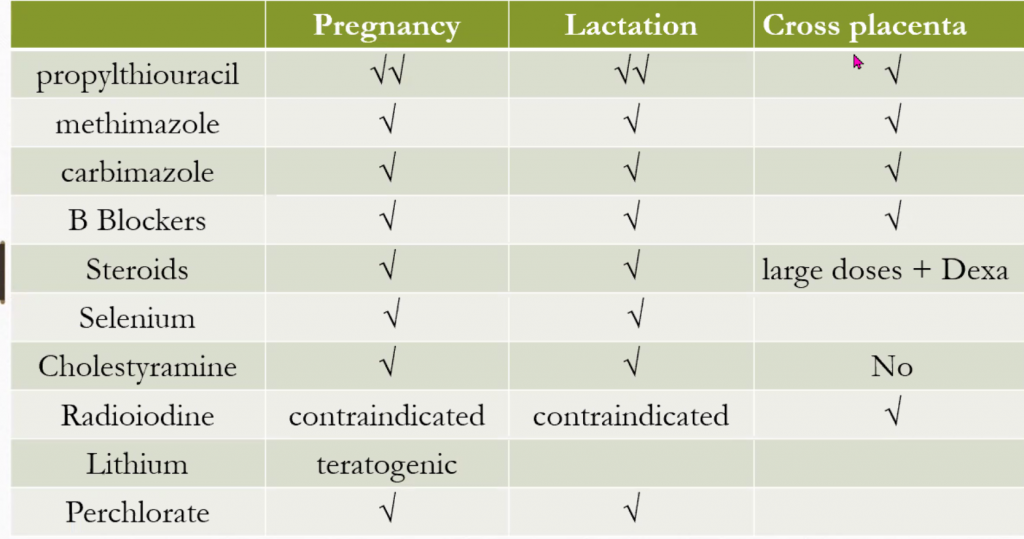
No specific guidelines for euthyroidism restoration before definitive treatment and authors have advocated the use of the following adjunctive therapies:
- Iopanoic acid
- Steroids (1 mg/ kg/ day)
- Potassium iodide solution (50—100 mg/d)
- Cholestyramine (4 g TDS)
- Lithium (400 mg twice daily)
- Steroids + Lithium / Steroids + Iopanoic acid
- Plasmapheresis
- Selenium
Practical points:
A dose of up to 90 mg CBZ was used in non-pregnant in literature (ATA recommendation in pregnancy 10 mg to 40 mg).
A dose of 150 mg MMI was used in literature in non-pregnant (ATA recommendation in pregnancy 5 mg to 30 mg)
A dose of 2000 mg PTU was used in literature in non—pregnant (ATA recommendation in pregnancy 100 mg to 600 mg)
The goal of treatment of hyperthyroidism in pregnancy with thionamides is to maintain free T4 in the upper normal range using the lowest possible dosage.
Combination of MMI + PTU was used in the literature
Beta Blockers use in pregnancy
Beta—adrenergic blocking agents, such as propranolol 10 – 40 mg every 6—8 hours may be used for controlling hypermetabolic symptoms.
The dose should be reduced as clinically indicated. In the vast majority of cases the drug can be discontinued in 2—6 weeks.
Long—term treatment with B-blockers adverse effects in pregnancy:
1. Small for gestational age 2.Foetal bradycardia 3.Neonatal hypoglycemia 4.One study suggested a higher spontaneous pregnancy loss rate when both – MMI & BB were taken together compared to patients receiving only MMI.