A case of persistent hypocalcemia
| HOSP # | MRN63985901 | WARD | Medical Ward |
| CONSULTANT | Dr. Heleen Vreede | DOB/AGE | 51 year Female |
Abnormal Result
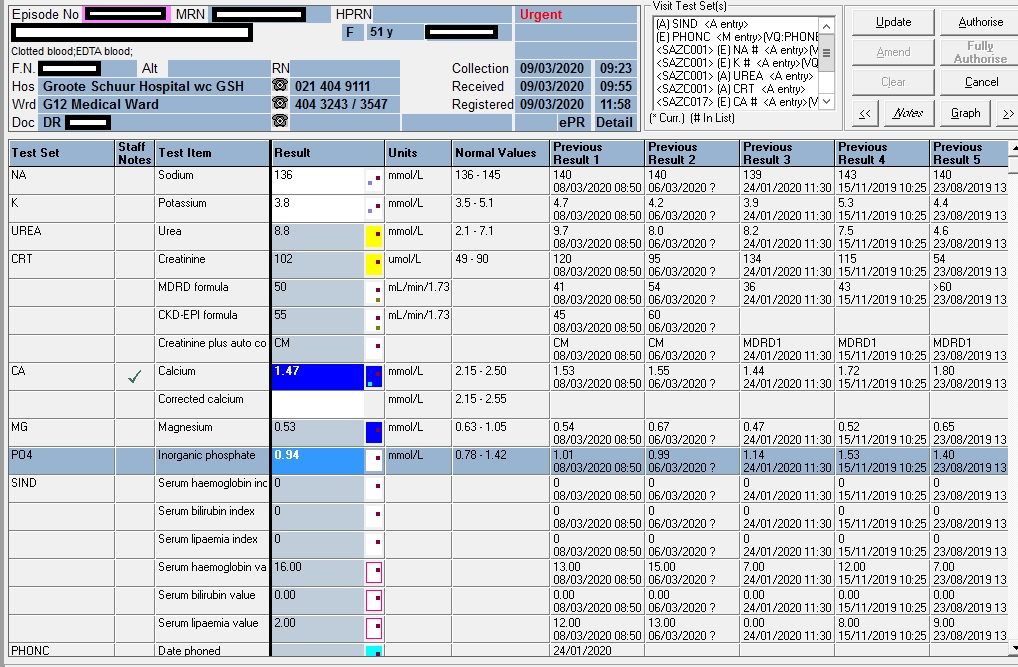
Total calcium of 1.47 mmol/L (2.15 – 2.50)
Presenting Complaint
The patient has been having persistent hypocalcemia despite supplementation with calcium.
History

Calcium: blue; Magnesium: orange; Phosphate: grey
Reference ranges are the horizontal lines without dotted markers
Examination
Not available.
The typical findings in a patient with true hypocalcemia (low ionised calcium) are
Trouseau’s sign
Chvostek’s sign
Laboratory Investigations
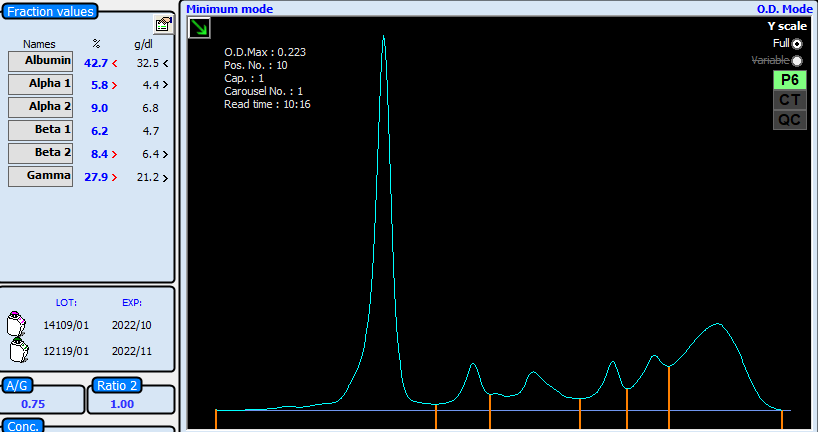
Arguably, the first important consideration in patients with low calcium is the albumin. The patient had a mean albumin of 12 g/L, significantly lower than normal (40-50g/L). Arguably, the calcium can be corrected with the well known Payne’s formula to then be 1.47 + (0.02 x (40-12) = 2.03 mmol/L:
Albumin-adjusted calcium (mmol/L) = total calcium (mmol/L) + 0.02 [40 – albumin (g/L)])
Payne RB, Little AJ, Williams RB, Milner JP. Interpretation of serum calcium in patient with abnormal serum proteins. Br Med J. 1973;4:643-646. DOI: 10.1136/bmj.4.5893.643. (View)
Measurement of serum intact parathyroid hormone (PTH) should be performed in all patients with hypocalcemia; it is the most valuable laboratory test for determining the etiology of hypocalcemia:
| 2019/11/15 | 2019/06/28 | 2018/08/03 | |
| PTH (pmol/L) | 21,8 H | 15,5 H | 25,8 H |
Vitamin D
| 09/09/2020 | 15/11/2019 | 03/08/2018 | |
| Total Vitamin D (25-OH VitD) | 20.5 nmol/L | 45.4 nmol/L | 23.2 nmol/L |
<30 nmol/L <12 ng/mL Deficient
30-50 nmol/L 12-20 ng/mL Insufficient
>50 nmol/L >20 ng/mL Sufficient
125-150 nmol/L 50-60 ng/mL Safe upper limit
Reference: Revised South African Clinical Guideline for the diagnosis and management of osteoporosis (NOFSA 2017), endorsing the institute of Medicine Dietary Reference intakes for calcium and vitamin D (2010). Note regarding conversion of units:
Divide result in nmol/L by 2.496 to convert to ng/mL
Multiply result in ng/mL by 2.496 to convert to nmol/L
Other Investigations
Anti-Tissue Transglutaminase antibodies: Negative: repeated 3 months apart, with sufficient IgA levels in the serum): 0.9 & 0.8 U/mL (EliA c/o: 6.9)
Anti-Gliadin antibodies: Equivocal: 7.8 & 9.6 U/mL (EliA c/o: 6.9)
Anti-endomysial antibodies: Negative
HLA-DQ2: Positive
HLA-DQ8: Negative
Final Diagnosis
Hypocalcemia likely due to malabsorbtion (telangiectasia stated by the clinicians).


Take Home Message
According to International guidelines the following association is expected for patients with Coeliac Disease:
Positive for HLA-DQ2 (HLA-DQA1*05, DQB1*02)
Positive for HLA-DQ8 (HLA-DQA1*03, DQB1*03:02)
Considering the fact that the albumin was high with an increased PTH, the calcium very likely was physiologically also low (bioactive Ca). The Payne’s formula also failed to correct the calcium to the normal reference range.