Section 12.1 – Declaration
Section 11.1 – Signature Page
Section 10.4 – FC Path (SA) Chem Exam Guidelines
Section 10.3 – Letter of Support to CMSA
Section 10.2 – FC Path (SA) Chem Part 1 Pass Letter
The Part I examination, with a pass mark of 50% comprises two 3-hour closed–book written examination papers with a subminimum of 50% (ie candidates need to attain 50% in each paper).
Paper 1 contain MCQs and short answer questions.
Paper 2 is a mixture of cases, calculations and OSPEs.
Current format for Part I
Paper 1: 75 MCQs and 75 Short answer questions (2 x 75 marks = 150 marks).
Paper 2: 8 cases (40 marks); 6 OSPE questions (30 marks); 6 calculations (30 marks); Total = 100 marks.
Pass Letter
Section 10.1 – Assessments
| Date | Type | Details |
| February 2019 | Written assessment | Paper 1: Essay questions (3hrs) Paper 2: Short answer questions (3hrs) |
| February 2019 | Written assessment | Paper 1: Essay and short answer questions (3hrs) Paper 2: Calculations, OSPE, Cases (3hrs) |
| February 2019 | FC Path(Chem) Part 1 Examination | Passed |
| June 2020 | Written assessment | Paper 1: Short answer questions, Cases, OSPE and Calculations |
| June 2020 | Written assessment | Paper 2: MCQs, Calculations, Cases, OSPE |
| June 2020 | Written Practical | Paper 3: Method Comparison |
| November 2020 | Written assessment | Paper 1: Short answer questions, Cases, OSPE and Calculations |
| November 2020 | Written assessment | Paper 2: MCQs, Calculations, Cases, OSPE |
| November 2020 | Written Practical | Paper 3: Method Comparison |
| November 2020 | Wet practical | Practical: total protein measurement with a method comparison scenario |
| June 2021 | Written assessment | Paper 1: Short answer questions, Cases, OSPE and Calculations |
| June 2021 | Written assessment | Paper 2: MCQs, Calculations, Cases, OSPE |
| June 2021 | Written Practical | Paper 3: Method Comparison |
| June 2021 | Wet practical | Practical: Mock practical exam and oral in a similar format to the CMSA exams |
| July 2021 | Part 2 Written assessment | Paper 1: Laboratory management: Short answer questions and long answer questions with MCQ’s – awaiting confirmation |
| July 2021 | Part 2 Written assessment | Paper 2: Pathophysiology: Short answer questions and long answer questions with MCQ’s – awaiting confirmation |
February 2019
June 2020 – Departmental Assessment
November 2020 – Departmental Assessment
Section 9.5 – Examples of Practicals
Section 9.4 – Standard Operating Procedures Followed
- Thin Layer Chromatography
- Plasma Free fatty acid analysis
- Deproteinising of samples
- Made PBS
- Manual Nucleic acid extraction
- PCR setup
- Restriction enzyme digest
- Agarose gel electrophoresis
- Checking gel
- DNA “clean-up” for sequencing
- Sanger sequencing with a capillary gel electrophoresis
Section 9.3 – Registrar Pack
Section 9.2 – Standard Operating Procedures Written
Section 9.1 – Methods and Practicals Performed
| METHOD | Performed | Observed | Neither |
| Basic spectrophotometry | Yes | ||
| Making solutions | Yes | ||
| Standard curve | Yes | ||
| pH measurement | Yes | ||
| Agarose electrophoresis | Yes | ||
| CZE | Yes | ||
| Serum osmolality | Yes | ||
| Glucose tolerance test | Yes | ||
| Sweat test | Yes | ||
| Creatinine-serum & urine | Serum – Yes | Urine | |
| Total protein | Yes | ||
| Bilirubin | X | ||
| Albumin | Yes | ||
| Enzyme assay | Yes | ||
| Enzyme kinetics | Yes | ||
| Blood gases | Yes | ||
| SDS electrophoresis | X | ||
| Solvent extraction | Yes | ||
| Thin layer chromatography | Yes | ||
| Radioimmunoassay | X | ||
| ELISA | Yes | ||
| Porphyrin Lab | Yes | ||
| Quality control | Yes | ||
| Method comparison study | Yes | ||
| Atomic absorption | X | ||
| PCR | Yes | ||
| DNA extraction | Yes |
Section 8.3 – Clinic Attendance
Lipid Clinic
Fridays February / March 2018
Prof David Marais & Prof Dirk Blom
We visited the Lipidology Clinic where Prof. Blom and Prof. David Marais sees the referred patients to the lipidology clinic. The patients seen here are referred generally due a suspected disorder of lipid or lipoprotein metabolism. It is a pity that more time cannot be spent mastering the clinical skills of lipidology in this clinic. The main indication for referral to this clinic is when pateints have a total cholesterol >7 mmol/L.
All patients initially gets a lipid electrophoresis (on agarose gel) to classify then according to the Fredericksen Classification (along with their lipogram).
I sat in with Prof. Marais on a few occasions where I learned the importance of thorough, structured history taking, including details of diet and exercise, as well as creating a pedigree to trace the family history. These are particularly important in dyslipidaemias which impacted significantly by both genetics and lifestyle.
Thorough history taking can assist to determine the degree to which each is contributing to the phenotype, and also helps with lifestyle counselling for disease
management. I also learned to appreciate the difference between a normal Achilles’ tendon and a thickened one, as well as examining for sterol deposits around the eyes (xanthelasma), in body folds and around joints (xanthomata).
Lipid Post-clinic case presentations
2018 – 2021
Prof David Marais & Prof Dirk Blom
Each of the clinicians that works in the lipid clinic presents the new patients that they saw, as well as the follow-up cases. The group discusses the new cases and comes to an agreement about the possibilty of familial hypercholesterolaemia, possible genetic background, and which drugs, at what doses, are most appropriate. Owing to Prof. Marais’ many years of experience, and ours being the only specialist lipid clinic and laboratory in the Western Cape, Prof. knows many of the families that are affected by FH and which mutations run in those families and in specific genetic pools within our population. A lot of clinical trials involve our patients, so we also hear up-to-date news on the latest developments in therapy.
Endocrine Ward Round
2018
Prof Dave Joel
A colleague and I asked whether we could join Prof Joel Dave on their Friday morning ward rounds. This was a very insightful experience as we could see the daily queries and consultations requested to review by an endocrinologist. We have learnt the importance of managing diabetes mellitus as it was the single most consulted endocrine disorder – also likely the most important non-commnicable disease on the rise. We have seen various cases in almost all the wards of the hospital, ranging from a patient with a recent radio-ablation of the thyroid, various cases of type 2 diabetes, a pregnant patient with diabetes for optimization of the insulin dose and a patient with Grave’s Disease. It was amazing to see with which care and confidence the clinicians handle the patients.
Endocrine Patient Presentations – Paper ward rounds
every Friday at 14h00
Prof Joel Dave, Prof Ian Ross
Every Friday, the adult and paediatric endocrinologists come together to discuss patients that present management or diagnostic dilemmas. The chemical pathologists and registrars are invited to assist with the diagnostic element and it’s another valuable opportunity for us to have closer contact with the patients. Sometimes, but rather rarely, we do go to see the patients at the bedside. I was involved in the discussion of a few of patients with a variety of fascinating diagnoses and dilemmas, including disorders of calcium metabolism, glycogen storage disease, complex cases of type I diabetes, including the issues that arise in adolescence.
There were cases of various types of Cushing’s syndrome, central and nephrogenic diabetes insipidus, disorders of sexual differentiation, lipid metabolism defects, growth hormone deficiency and acromegaly. What I enjoyed about these paper rounds is that we would become thoroughly involved in the discussions and decisions about the next diagnostic step and I feel we really added value in real-time. It was much better than consulting over the phone, because we were able to look at laboratory and imaging results together, and hear all the questions and discussions. When we consult over the phone, we miss out on a lot of that background discussion and problem-solving.
Paeds Endocrine Clinic – Red Cross Children’s Hospital
Monday Mornings
Dr. M. Carrihill and Dr. A Ramcharan
I visited the endocrine clinic a few mornings when the endocrinologists had interesting cases. I have become the “go-to” person for organizing urinary steroid profiles in the Western Cape region, with the contacts I have made with laboratorians at the WADA-SADoCoL laboratory in the Free State. Sometimes when children with disorders of sexual differentiation presents, I am consulted on the possibility of sending these special tests for analysis. After a few analyses, we realized that these tests are only useful to confirm 5-alphareductase deficiency in older children, using the testosterone:dehidrotestosterone ratios. The sensitivity of the other urinary analytes, present in low amounts in pediatric urine, namely 5-alpha: 5-beta THF and androsterone : aetiocholanolone was unfortunately not good enough.
Unfortunately it is was not easy to get the time to regularly attend these meetings as we also have our regular Journal club and staff meetings on Mondays.
Dynamic Tests
- OGTT for diabetes
- OGTT for acromegaly
- Clonidine stimulation test
- hCG stimulation test
- Water deprivation test
- Overnight fast for hypoglycaemia
- Low dose dexamethasone suppression test
- Synacthen stimulation test
These dynamic tests are fairly often requested at our laboratory. The chemical pathology registrar on call is responsible for taking the clinician’s call, organizing that the clinician is well-informed of which samples should be taken and also to liaise with the clinician if results need to be phoned out, or if additional samples are necessary. We are also responsible to obtain the history and clinical scenario and ensure that the appropriate tests are done, at the appropriate time. These tests, although usually requested by endocrinologists at our laboratory, are often requested by younger clinicians, or clinicians not fully aware of the caveats with doing these tests. To give an example, we have had a few requests for a ADH (vasopresssin) lever. It is unfortunate that we do not have the option to measure this analyte, neither would it likely be very specific to disease due to the short half life. Nonetheless it is then our responsibility to inform the clinician of the water deprivation test and assist with a suitable protocol to perform the test. Similarly, an overnight fast for hypoglycaemia is usually planned well in advance with us on board with the advisements on minimum sample volumes and ensuring the correct tests be done under the correct conditions and sent to the correct laboratory.
Near-Patient Tests
Sweat Test
The whole procedure of the sweat test from start to finish requires a fair amount of time, so I wasn’t able to follow many of these patients. I did spend a day with our technologist at Red Cross Children’s hospital, Ms Sandy Kear, when we did sweat tests on 4 patients that day. One out patient and three patients in the ward. It is interesting that such a laborious test remains the gold standard for diagnosing cystic fibrosis. One expects the collection of sweat and manual handling of samples to result in significant variability in results, but clearly the differential in results between normal and abnormal is sufficient to withstand this variability. I was also involved in repairing one of the newer iontophoresis machines for the Macroduct, a variation on the Whatmann paper sweat collection method. See my case of 3D printing in the laboratory for more information. I was also involved in the EQA for the chloride measurement. See Record of Rotations for more information.
Selective arterial calcium stimulation test (Calcium gluconate infusion test)
Patient experienced hypoglycaemic episodes associated with elevated insulin. CT scan could not localise a tumour. SACST was performed. This procedure is much simpler than a BIPSS in terms of administration, but with all of these procedures, great skill is required in the correct placement of the catheter to obtain valid results. The results showed an elevated insulin response to calcium infusion in the superior mesenteric artery and splenic artery, but minimal response in the gastroduodenal artery. This suggested an adenoma in the body/tail of the pancreas. Ultrasound-guided surgery was planned, with the intention of a local resection if possible.
Bilateral adrenal vein sampling
Since I have had exposure to the Calcium stimulation test, and due to space restrictions, I could unfortunately not see first-hand this procedure. I was however well informed by the single colleague who was allowed into the CathLab to help with specimen logistics, about the procedure. See my short case on hyperaldosteronism for more details. There were two of these cases on one day. It did appear that the interventional radiologists could not adequately canulate the right adrenal vein, a commonly encountered problem.
Bilateral inferior petrosal sinus sampling
Even though also not directly involved in the theatre, I’ve been informed by colleagues various times about this procedure which has been done just before I arrived in the department. The patient was diagnosed with Cushing’s disease via a private laboratory. Colleagues assisted by preparing all the tubes and ice before the procedure, and were involved in the coordinated collection of samples at the different time points from each anatomical location. ACTH secretion was stimulated with ddAVP, but the ratio of central to peripheral ACTH secretion was not diagnostic of pituitary Cushing’s disease. Therefore, ectopic ACTH syndrome was diagnosed. This appears to be a very involved procedure and requires the presence of a multidisciplinary team to ensure everything is done correctly. After the Calcium stimulation test I have assisted Prof. Beningfield with, I wish I could see this procedure from him too – one of the legends of the radiology department.
Section 8.2 – Evidence of Clinical Case Learning (Short Cases)
Section 8.1 – Evidence of Clinical Case Learning (Long Cases)
Section 7.7 – Laboratory Business Plan Presentation
At the Laboratory Management course, attended virtually at the University of Stellenbosch, our task was to give a presentation of our business plan.
The task was as follows:
You are the head of an accredited academic pathology laboratory (you have to annually sustain its accreditation status) at a large 1000 bed teaching hospital in one of the developing countries in Africa. You are in charge of providing laboratory tests to a population of 3 million people who are spread out (large urban and rural pockets). In addition to providing a comprehensive tertiary quality service you also teach undergraduate students and train postgraduate specialists. One of your other duties is to carry out research and ensure research outputs that are of international standard as well as supervise postgraduate students and apply for large grants to support these projects. Your academic laboratory closely works with the medical faculty and the Medical Research Council of your country. There are 2 private pathology laboratories who also compete with your laboratory for proving a service. You are also expected to provide community services and cover the province in which your laboratory is located. As part of the 5 year service and academic plan you have been asked to develop a strategic plan for your department for the next five years. Your team will present this plan for discussion and approval
You are divided into 5 groups. Each group will make a presentation at the end of the course on Thursday afternoon (05/11/2020) and your presentation will be assessed. Each presentation will last for 20 mins and will take the form of a group presentation in which all members of your group will present various aspects of the strategic plan. Group discussion time must be used for this activity.GROUP ASSIGNMENT, DISCUSSION AND FINAL PRESENTATION
We were a multi-disciplinary team, consisting of 2 Chemical Pathology registrars, a Hematology registrar as well as a Histopathology registrar. This task was taken on with great enthusiasm and the presentation received positive feedback. See downloadable pdf version of the presentation below:
Section 7.6 – Mmed
Please see addenda or download from the link below the chromatograms:
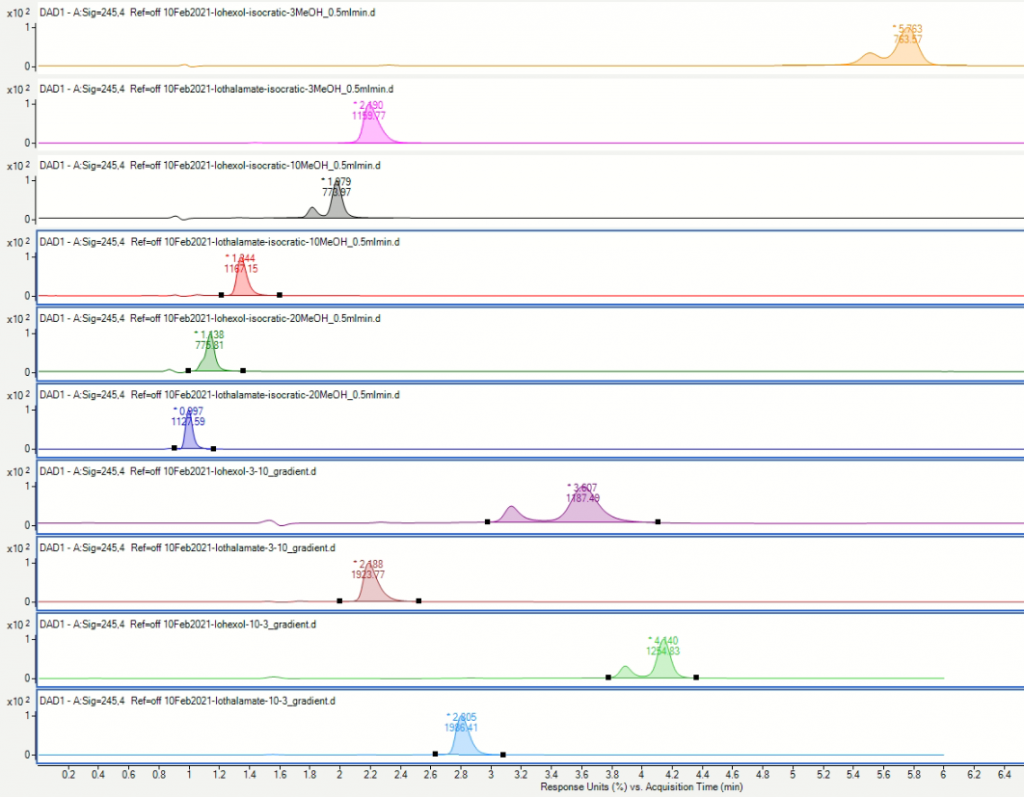
Section 7.5 – Tasks
Various auditorial and management tasks has been performed in the laboratory throughout my 4 years. These include but are not limited to:
- ChemHelp – A script (program) which helps reviewers with the signing of blood results. It consists of various hotkeys, hotstrings and a few buttons which has evolved as our “right hand” when reviewing blood results at Groote Schuur Hospital NHLS. On face value it appears as a small petit application which runs alongside TrakCare, but the time saving is likely to be quite significant. This will likely be a nice additional audit – another project which could be taken on later. Unfortunately the descriptions of the functions of this script goes beyond the scope of this section, so please refer to Section 9.2.4 – ChemHelp SOP or visit: github.com/dietervdwes/ChemHelp (hyperlinks unfortunately are non-functional in a combined and printed PDF).
2. Data extraction tasks for projects and audits – Apart from being involved in an audit in our laboratory, I have been involved in assisting with data extraction via TrakCare’s Results Listing interface. Additionally, I have written a JavaScript extraction tool which has been used (and are being used) in a few studies currently. I have also incorporated into ChemHelp (see above) an automation tool for data extraction via TrakCare’s Results Listing interface. See Section 9.2.
3. Management and advisement of some pre-analytical aspects are sometimes required. As registrars we need to advise and help improve pre-analytical staff’s knowledge through assistance with routine test request queries. We also have a programme where the registrar’s do what we call a “Tech Talk”. These are mainly aimed at technologists but I have also given a talk to an audience including pre-analytical staff on aspects like sample delay, hemolysis and the effect thereof on HIL-indices.
4. Setting up of a referral system – I have set up a referral system for urine steroid profiles in our laboratory. I was involved in identifying the referral lab, setting up the billing codes on TrakCare, with Dr. Heleen Vreede and setting up the canned comments for recording of the results. Invoicing also caused some problems which we had sort out.
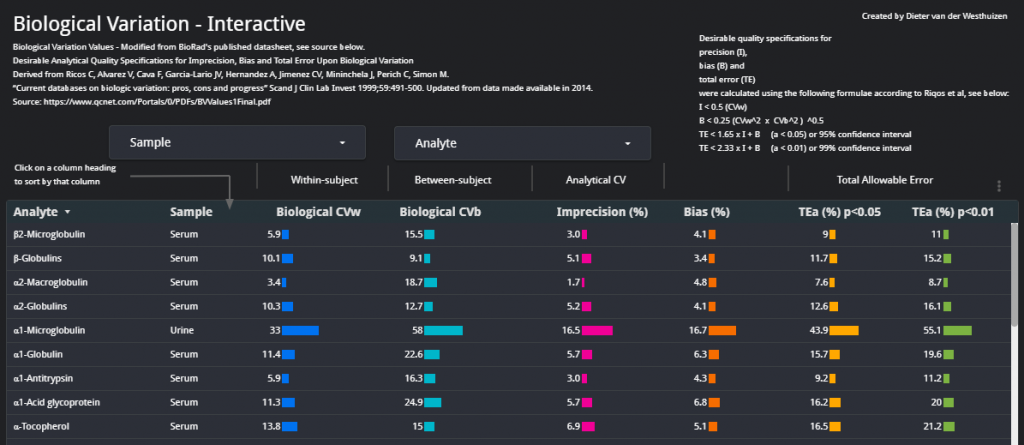
5. TEa Dashboard – I have taken the data from the Riqos et al. study on Biological Variation (updated in 2014) and made an interactive dashboard which we can use when reviewing QC. This dashboard assists in quickly viewing the data visually in a quick-to-access Google Data Studio Dashboard.
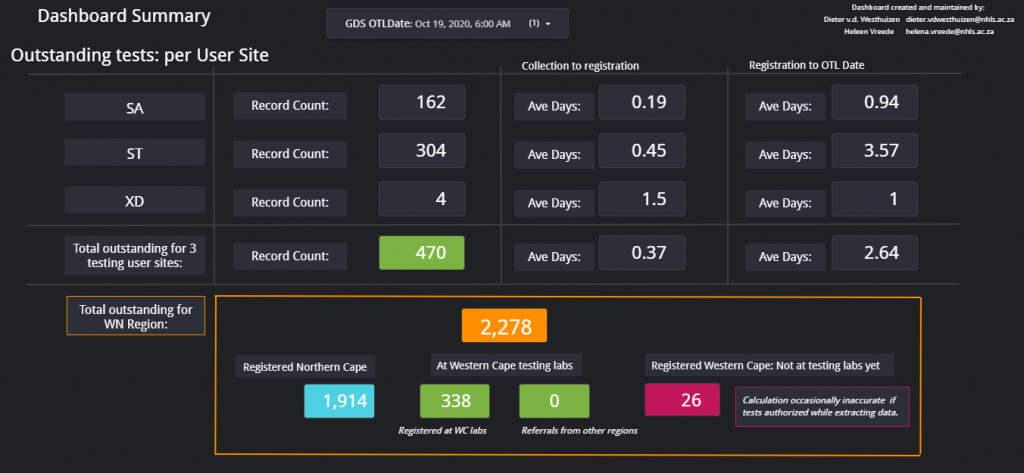
6. COVID OTL Dashboard – During the first COVID Lockdown most of the COVID PCR labs in South Africa were swamped with samples and couldn’t easily keep up with the testing volume. The Department of Health needed info of the back log of samples to be tested. Additionally, the Western Cape Area manager needed information of where the hold-up of samples were in the province. I was contacted as I had some (albeit limited) experience with databases and dashboards. The aim was to make a dashboard which could effectively track delayed samples, count them between centres and also determine where possible hold-ups were. With Dr. Heleen Vreede we set up automated data extractions on TrakCare (every 12 hours at 07h00 and 19h00) and made it such that the data extractions could be loaded onto a Google Sheets Database where Google Data Studio would get the data to calculate the respective counts and average outstanding times. This was done for the 3 COVID testing laboratories in the Western Cape at the time: Groote Schuur Hospital laboratory, Tygerberg laboratory and Green Point laboratory. There were various functions and calculations in this dashboard, with 4 large tables in a database. The dashboard also had various pages, each of which could be used for a specific query. Although not entirely focussed on Chemical Pathology, this project has learned me much about data science in general. As an example, two screenshots of the first page are shown below. The dashboard can be viewed at: WC OTL Dashboard – tinyurl.com/COVID-WC-OTL
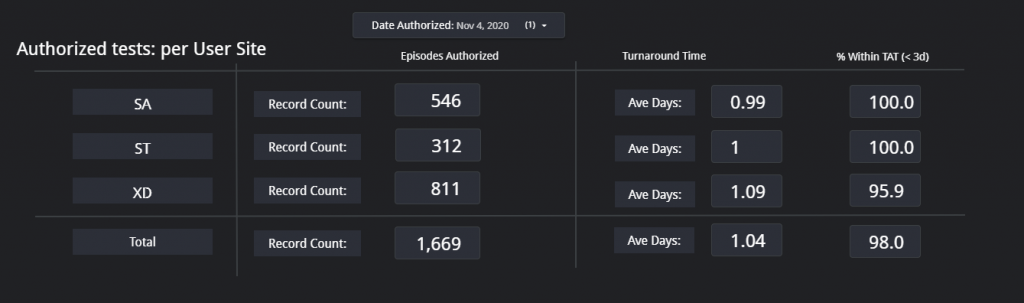
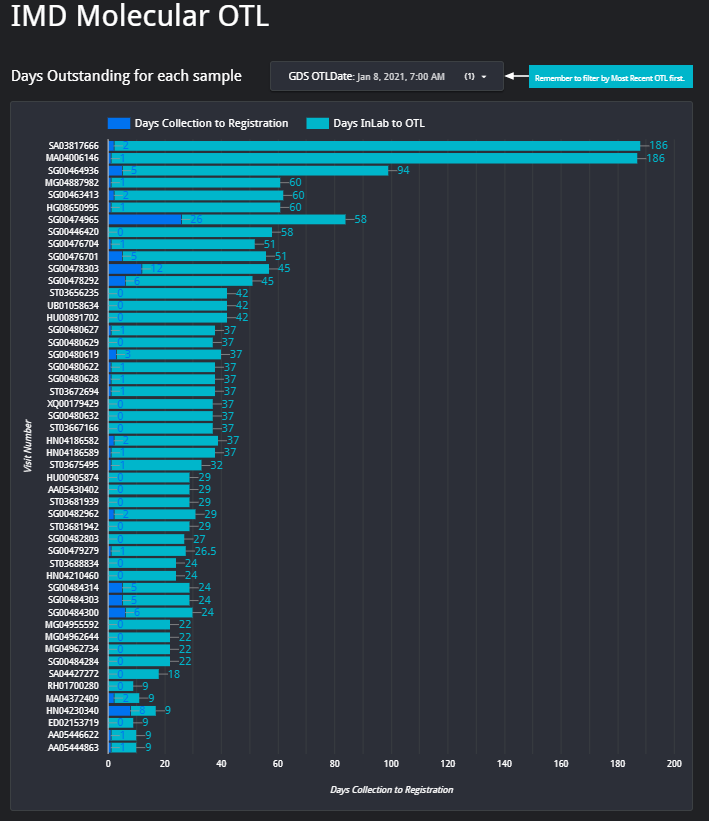
7. IMD OTL Dashboard – From above Dashboard, I was then approached by Surita Meldau, our IMD Genetics Laboratory Head to assist with the IMD Genetics’ lab OTL’s to try come up with a solution to easily track the most outdated samples for the planning of which samples to prioritize each week. We then implied the same principles of above COVID dashboard, into this IMD OTL Dashboard. The result was an interactive portal where we can view information such as patient name, surname, requesting hospital and type of genetic test, all on one platform. The reports are not generated 12-hourly as in the COVID dashboard, but weekly every Friday morning.
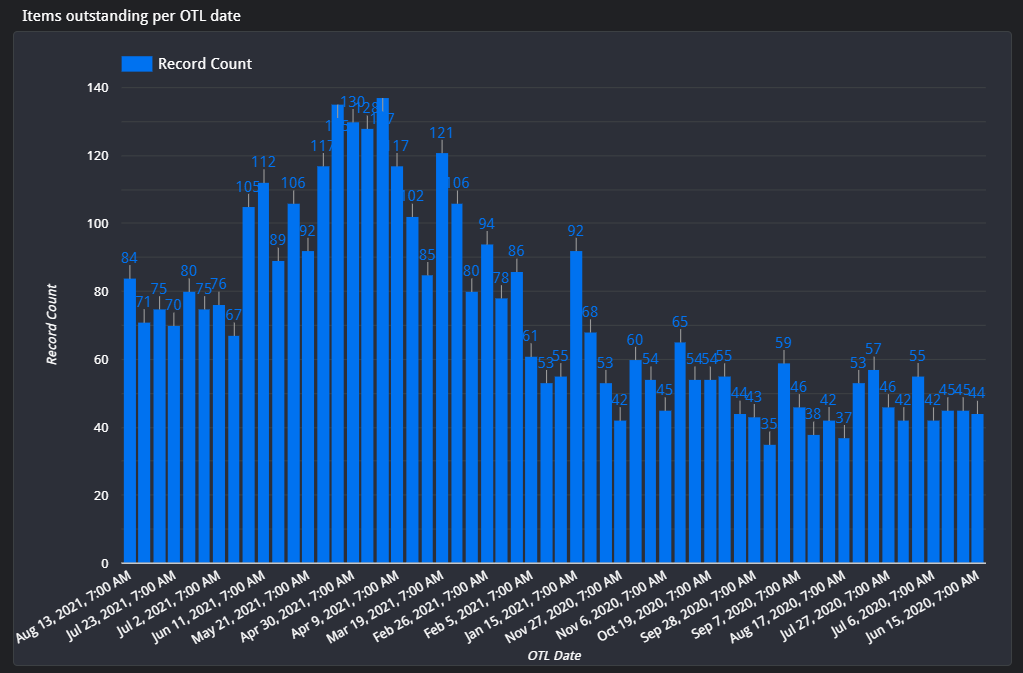
8. Transcribing results to TrakCare from CSV in an automated way – With the rapid ramp-up of COVID PCR and antibody testing, there were a few problems initially, which ranged from TrakCare database problems when the new test sets were created, billing problems and analyzer-to-LIS-interface problems. A few times I needed to assist with a few thousand results which needed to be removed and re-tranmitted or entered onto TrakCare. Soon I wrote a script which we could run on a computer, and later on a few computers at once to do the data entry onto TrakCare in an automated way from a CSV results file. This script has no SOP, but is hosted on Github is the need arrises again to use it. It is customized to the applicability at every instance.
Section 7.4 – Method Verification
Introduction
A new analyzer (Diasorin Liaison) was aqcuired by the Virology Department at GSH.
In summary: since the Diasorin Liaison has the capability to do various chemistry analyses too, we were able to “piggy back” on the analyzer’s acquisition and install / setup various chemistry analytes too.
We have decided to introduce:
- Aldosterone
- Renin
- IGF-1
- Growth Hormone
- C-peptide
I was given the task to specifically concentrate on aldosterone as method verification.
Points which will be discussed:
- Rationale for implementation of Aldosterone and Renin
- Medical decision limits
- EP15-A3: 5×5 method for precision (and accuracy) determination
- Method comparison study
Rationale for implementation of Aldosterone and Renin
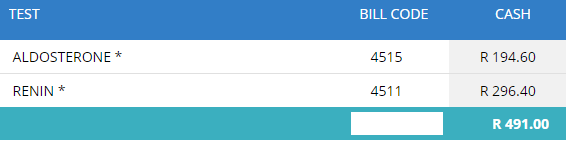
Our previous practice was to send aldosterone and renin results to a private laboratory (PathCare).
Virology had planned to budget for a new analyser to run a few of their Virology serology assays. Their test volumes, for the assays on their analyser hasn’t been high enough to adequately motivate for this new analyser.
The auto-analyser which we use (Roche COBAS 6000), doesn’t have a repertoire of Aldosterone and Renin.
Since we had already sent roughly 30 samples for both aldosterone and renin to Pathcare per month, this was a great opportunity to partner with the Virology department in a mutual tender for an instrument which could run their serology assays and our Aldosterone and Renins. Additionally we added some other tests to the repertoire for tender of tests which we were also sending to Pathcare: IGF-1 and hGH.
Together with the anticipated virology serology tests, the total test volume was such that we could motivate to the business manager at GSH for a “break-even” scenario with regards to Cost vs. Income.
The tender was then awarded to the company supplying the Diasorin Liason analyser (as a placement option for 5 years), upon which I was given the task of doing the method verification for aldosterone.
The prior CLSI EP15 A2 document described the following:
Clinical and Laboratory Standards Institute (CLSI) document EP05-A2 describes the protocols for determining the precision of a method. The precision of a method should be tested at at-least two levels; each run in duplicate, with two runs per day over 20 days. It also describes the protocols that should be undertaken by the user to verify precision claims by a manufacturer. Precision claims by a manufacturer should be tested at at-least two levels, by running three replicates over five days.
Excerpt – CLSI EP15 A2 2
Medical decision limits
The medical decision limits chosen were:
EP15-A3: 5×5 method for precision (and accuracy) determination
In the NHLS, a method verification currently happens according to guidelines as per CLSI EP15-A3 document: “User Verification of Precision and Estimation of Bias” provides a simple experimental approach to estimate a method’s imprecision and bias. 1
The objective is to determine if the laboratory precision performance of repeatability and within-laboratory imprecision are in accordance to the manufacturer specification claims.
Aldosterone was performed on the Diasorin Liason. One QC lot was obtained for QC of the assay after maintenance and another QC lot (with a different assigned value) was used as the testing material. Quality control (QC) levels (L1: 5.25; L2: 21.10 ng/dL) (Bio-Rad Laboratories, Irvine, CA) were used. Each QC level was measured before and after instrument. Five replicates of the testing material was done per run, one run per day, and during 5 days. Imprecision estimates were calculated by one-way analysis of variance using Microsoft Excel on a template sheet developed previously by Dr Fierdoz Omar. Estimated imprecision was compared to the manufacturer’s stability claims and desirable imprecision specifications based on biological variation.
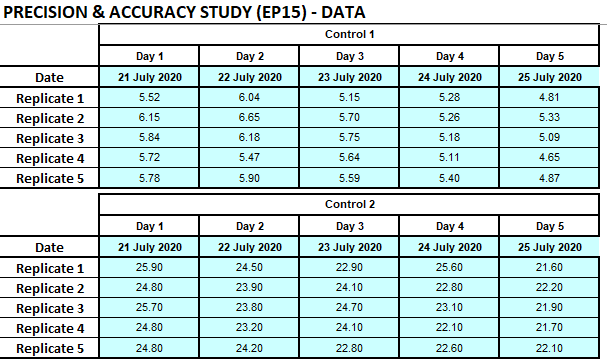
Result
See below the attached verification report:
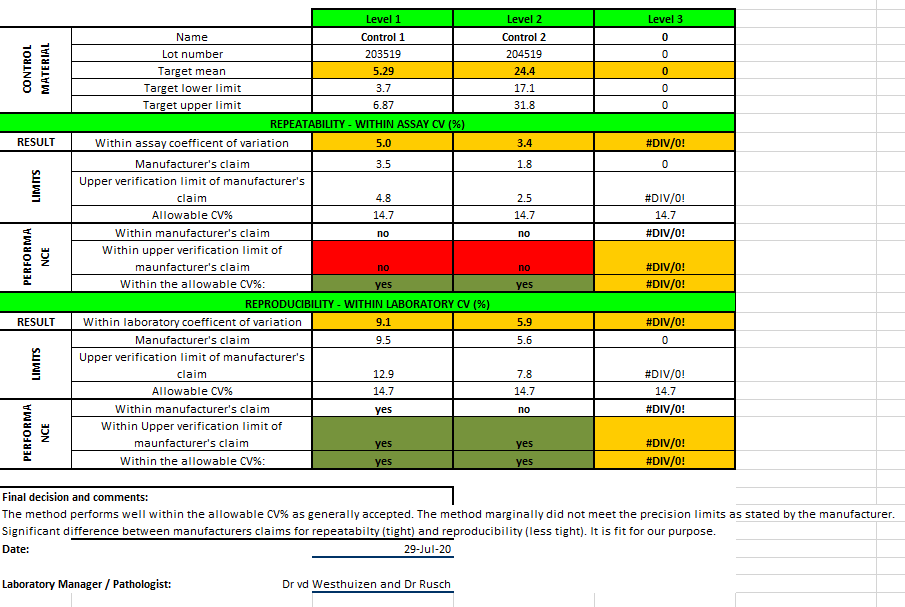
The full report can be downloaded here:
Conclusion
The aldosterone assay was found “fit for purpose” although not meeting all the manufacturer’s claims.
The literal description of “fit for purpose”: (of an institution, facility, etc.) well equipped or well suited for its designated role or purpose.
This assay, even though not meeting the manufacturer’s claims, performs well within the published “Desirable Analytical Quality Specifications for Imprecision, Bias and Total Error Upon Biological Variation”.
In the process of this project and every day IQC, I have also developed an interactive portal with the desirable analytical quality specifications as per Ricos et al.: TEa Dashboard
References
- Jose Jara Aguirre, MD, Karl Ness, MLS, Alicia Algeciras-Schimnich, Application of the CLSI EP15-A3 Guideline as an Alternative Troubleshooting Tool for Verification of Assay Precision, American Journal of Clinical Pathology, Volume 152, Issue Supplement_1, October 2019, Page S88, https://doi.org/10.1093/ajcp/aqz117.007
- Chesher D. Evaluating assay precision. Clin Biochem Rev. 2008 Aug;29 Suppl 1(Suppl 1):S23-6. PMID: 18852851; PMCID: PMC2556577, https://www.ncbi.nlm.nih.gov/pmc/articles/PMC2556577/
- Desirable Analytical Quality Specifications for Imprecision, Bias and Total Error Upon Biological VariationDerived from Ricos C, Alvarez V, Cava F, Garcia-Lario JV, Hernandez A, Jimenez CV, Mininchela J, Perich C, Simon M. “Current databases on biologic variation: pros, cons and progress” Scand J Clin Lab Invest 1999;59:491-500. Updated from data made available in 2014.Source: https://www.qcnet.com/Portals/0/PDFs/BVValues1Final.pdf
Section 7.3 – Audit
The following poster has been presented at a Congress Pathcape in 2018 as a result of this audit. It also summarizes the work that has been done.
Download
—————————————————————————————————–
Laboratory Audit
Overcoming incomplete laboratory request forms:
Is an updatable online database of clinician contact details the answer?
DJ van der Westhuizen, J Cole, R Dalmacio, EJ Gantana, JA Rusch, HW Vreede
Division of Chemical Pathology, University of Cape Town & Groote Schuur Hospital, National Health Laboratory Services
Background
As a result of omission of contact details on laboratory request forms, laboratory personnel frequently spend excessive time attempting to reach responsible clinicians regarding patient results. Poor completion of request forms was previously established in our laboratory as well as at the nearby Tygerberg Hospital NHLS.1 An online web form was created in our laboratory that allows clinicians to update the contact details linked to their professional registration number
Objective
This audit is intended as a proof of concept that the proportion of clinicians that are readily contactable would significantly increase should a system that links professional registration numbers with valid contact details be implemented.

Method
All request forms for the Core Laboratory of Groote Schuur Hospital (encompassing chemistry, haematology, immunology and virology) over a two-day period were scrutinised to determine the proportion of clinicians that provided their contact details and HPCSA registration numbers. The completeness of other clinically and analytically relevant fields was also recorded. Request forms received from all hospitals and clinics in our catchment zone were included.
Results
A total of 2301 forms were analysed.
- Personal contact – and pager numbers were absent on 81.6% of forms.
- The ward or clinic contact number was absent on 64.3% of forms.
- Of the 34.4% of forms which exhibited an HPCSA registration number, 41.1% lacked a personal contact number and pager number.
- Of all forms from local clinics, 46% provided no contact number.
- Specimen collection time was absent in 24.7% of forms.
- Collection date was absent in 3.5% of forms
- Clinical details was absent in 34.5% of forms.
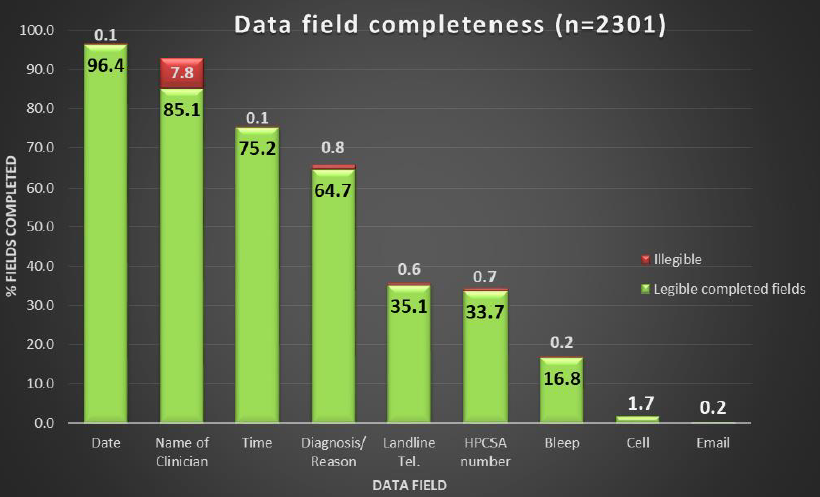
Conclusions
The vast majority of clinicians do not provide personal contact details for the communication of critical laboratory results. This audit illustrates that a significant improvement may be made were clinicians able to maintain their contact details on an online form that links these details with their professional registration number. Such a form has been created and awaits approval by the NHLS executive. Should it be approved, it will be made available to clinicians in the Western Cape who are served by the NHLS. After sufficient time has elapsed, an audit can be undertaken to monitor the effect of this intervention.
References
1. Nutt L, Zemlin AE, Erasmus RT. Incomplete laboratory request forms: the extent and impact on critical results at a tertiary hospital in South Africa. Annals of clinical biochemistry. 2008 Sep;45(5):463-6.